On December 31, 2025, The Supreme People’s Court upheld the Beijing IP Court’s decision, meaning that the semaglutide patent remains valid and will expire on March 20, 2026. Novo Nordisk announced this positive news via a press release on the day the case was decided. The Chinese Ministry of Commerce has also confirmed this decision on their website. This case is certainly positive news for those in the biopharmaceutical industry thinking of developing their drugs in China. Additionally, if you’ve been following this case, you’ll know that the validity of the patent came down to whether the Court would accept post-filing data demonstrating semaglutide’s surprising pharmacokinetic effects in animal models.…
-
- China, China Patent Office, CNIPA, data, Invalidation, Patent, Patent Law, Patent Re-examination Board, Patentability, Pharma, Support Requirements
How to Protect a Crystal Form (Polymorph) Patent in China
Crystalline forms are critical to pharmaceutical patents, offering extended protection for improved stability, bioavailability, or manufacturability. However, securing such patents in China has grown increasingly difficult due to the China National Intellectual Property Administration (CNIPA)’s strict patentability criteria. Unlike the U.S. or Europe, where structural novelty or problem-solving utility may suffice, China demands quantifiable evidence of superiority over prior art forms and rejects patents based on routine screening alone. Recent decisions, like the invalidation of fruquintinib Crystal Form I, highlight common pitfalls: insufficient comparative data, incremental technical effects, and failures to preempt obviousness challenges. With China’s pharmaceutical market surging and secondary patents under heightened scrutiny, companies must strategically align their IP strategies…
-
Beijing IP Court Reverses CNIPA Decision and Upholds Ozempic® semaglutide patent in China as VALID based on Novo Nordisk’s Post Filing Data
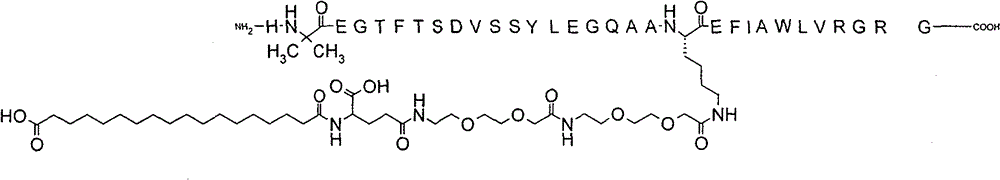
Recently, all eyes have been on China as the fundamental patent covering semaglutide, the active ingredient in Ozempic® and Wegovy®, will expire on March 20, 2026. It goes without saying that generics are ramping up bigtime in China (and also around the world), preparing to manufacture and sell this blockbuster drug to one of the biggest markets in the world. Any shortening of the patent term for this key semaglutide patent in China could cause an immediately shift in the Chinese Ozempic market (not to mention directly impacting Novo Nordisk). Novo Nordisk’s Semaglutide Patent in China On September 5, 2022, the China National Intellectual Property Association (China’s patent administrative office,…