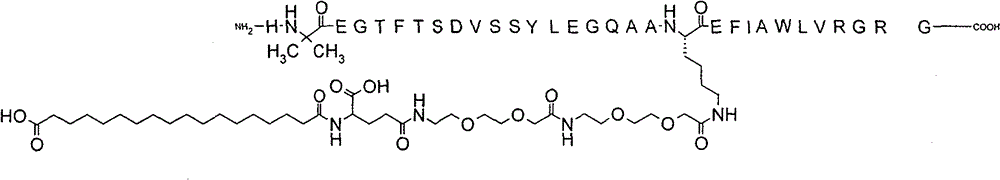
On December 31, 2025, The Supreme People’s Court upheld the Beijing IP Court’s decision, meaning that the semaglutide patent remains valid and will expire on March 20, 2026. Novo Nordisk announced this positive news via a press release on the day the case was decided. The Chinese Ministry of Commerce has also confirmed this decision on their website. This case is certainly positive news for those in the biopharmaceutical industry thinking of developing their drugs in China. Additionally, if you’ve been following this case, you’ll know that the validity of the patent came down to whether the Court would accept post-filing data demonstrating semaglutide’s surprising pharmacokinetic effects in animal models.…
-
- AI, China, China Patent Office, CNIPA, Inventions, Inventiveness, Patent, Patent Re-examination and Invalidation Department, Patent Re-examination Board, Top 10 IP Case
From Faces to Football Fields: How Applying AI Models to Different Application Fields Can Prove Inventive in China
As AI models become versatile and adaptable across multiple contexts and industries, questions about inventive step sit at the heart of patent examination in China. The 2023 Patent Examination Guidelines (hereinafter referred to as ‘the Guidelines’) included several sessions dedicated to explaining how inventive step should be examined for AI-related inventions, featuring examples related to AI algorithms, big data, and user experience1. In the re-examination of Beijing ByteDance Network Technology Co. Ltd.’s application entitled “Method and apparatus for processing an image” (Application No. 201810734681.2)2, which was recognized as one of the Top 10 Re-examination Cases of 2025 by the CNIPA, the CNIPA focused on the following issue: when an existing…
- China, China Patent Office, CNIPA, data, Invalidation, Patent, Patent Law, Patent Re-examination Board, Patentability, Pharma, Support Requirements
How to Protect a Crystal Form (Polymorph) Patent in China
Crystalline forms are critical to pharmaceutical patents, offering extended protection for improved stability, bioavailability, or manufacturability. However, securing such patents in China has grown increasingly difficult due to the China National Intellectual Property Administration (CNIPA)’s strict patentability criteria. Unlike the U.S. or Europe, where structural novelty or problem-solving utility may suffice, China demands quantifiable evidence of superiority over prior art forms and rejects patents based on routine screening alone. Recent decisions, like the invalidation of fruquintinib Crystal Form I, highlight common pitfalls: insufficient comparative data, incremental technical effects, and failures to preempt obviousness challenges. With China’s pharmaceutical market surging and secondary patents under heightened scrutiny, companies must strategically align their IP strategies…