Each year the CNIPA publishes its Top Ten Patent Re-examination and Invalidation Cases for the previous year. These cases are meant to be guiding cases, showcasing exemplary real-world decisions that clarify certain aspects of the law. Today we’ll be sharing about one of the Top 10 Invalidation Cases in 2024 involving Microsoft (China) Co., Ltd. petitioning to invalidate a patent by Newman Infinite Inc. in the field of human-computer interaction (HCI). Case summary The patent claimed methods and devices for manipulating a touch screen user interface that could help differentiate between various touch targets, preventing undesired interactions with non-target elements. The patentee used a self-defined term “clutch user interface element”,…
-
-
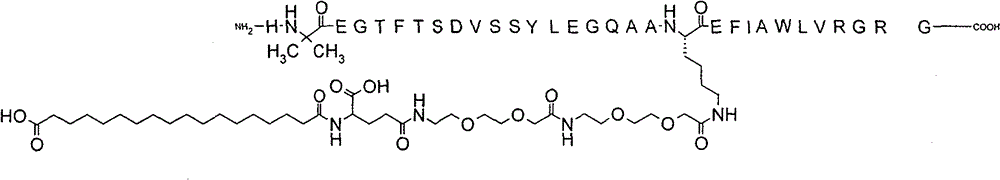
Beijing Supreme People’s Court Upholds Novo Nordisk Semaglutide Patent in China based on Post-Filing Data
On December 31, 2025, The Supreme People’s Court upheld the Beijing IP Court’s decision, meaning that the semaglutide patent remains valid and will expire on March 20, 2026. Novo Nordisk announced this positive news via a press release on the day the case was decided. The Chinese Ministry of Commerce has also confirmed this decision on their website. This case is certainly positive news for those in the biopharmaceutical industry thinking of developing their drugs in China. Additionally, if you’ve been following this case, you’ll know that the validity of the patent came down to whether the Court would accept post-filing data demonstrating semaglutide’s surprising pharmacokinetic effects in animal models.…
-
Thin Disclosure but “Bulletproof” Protection: How a Salt Form Patent was Upheld in China Despite Minimal Data
In China, innovative drug companies typically rely on their initial composition of matter patents to block out competitors. Other “follow-on” IP, such as those directed towards formulations, polymorphs, and salt forms, are often regarded as less likely to survive invalidation challenges, especially in China where the standard of “person skilled in the art” is so high. Having said that, we share below today a positive counter example case in which a salt form of a JAK inhibitor survived invalidation. This case is one of CNIPA’s Top Ten Patent Re-examination and Invalidation Cases for 2024 (published in the 2025 list). These cases are meant to be guiding cases, showcasing exemplary real-world…