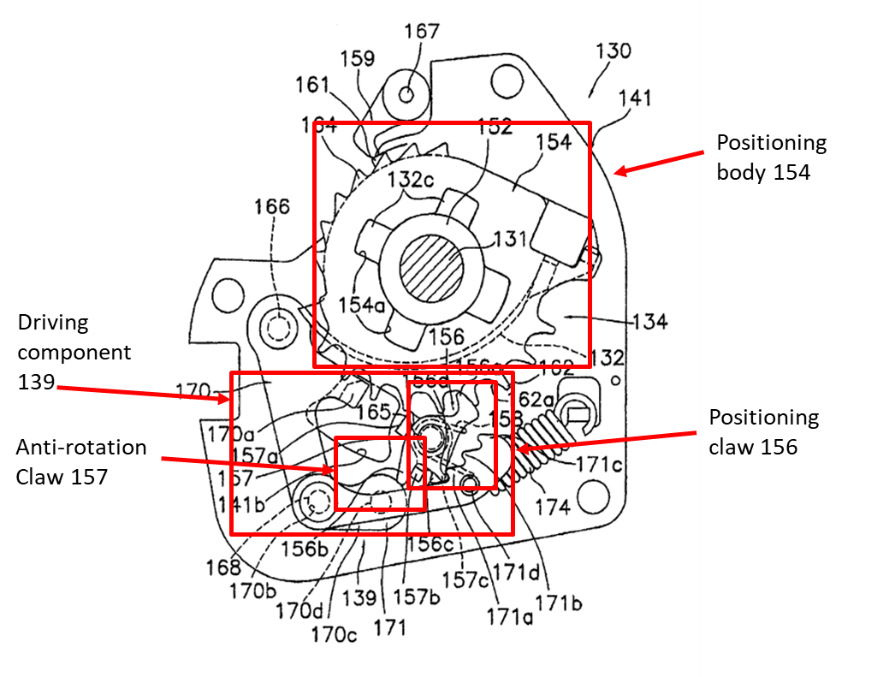
2019 China’s Top 50 Representative IP Cases Shenzhen Weibang Technology Co. Ltd v. Li Jianyi & Shenzhen Yuancheng Intelligence Equipment Co. Ltd (2019最高法民申6342号) Each year in China there are hundreds of disputes over patent ownership. A significant part of them are related to “Service Inventions”, which are defined in Article 6 of the Chinese Patent Law as any invention made by an employee (1) in the course of performing the employee’s duties or (2) mainly by using the materials or technical means of the employer. Under Chinese patent law, these inventions automatically belong to the employer. No separate assignment or employment agreement needs to be in place. Article 12 of…
-
-
China Top 50 Case of 2019: How a Functional Feature is Construed Greatly Impacts Protection Scope – Shimano KK v. SENSAH
Every year in the spring The Supreme People’s Court of PRC announces its “Top 10 Intellectual Property (IP) cases” and “50 typical IP cases in Chinese courts” of the year. Although not having precedential weight, these cases serve as a reference and guide people’s courts at all levels regarding their intellectual property trials. It’s always interesting to see which cases get chosen, since it reflects what concepts and messages the Courts want to highlight to the public. Below, we highlight one of the 50 typical IP cases. Shimano KK v SENSAH Smart Sports Equipment Co., Ltd[(2019) Supreme Court Minshen No.5466] In China, cases can turn completely on a single determination…
-
Polymorph Patents in China: What is the Standard for Inventiveness for New Crystal Forms?
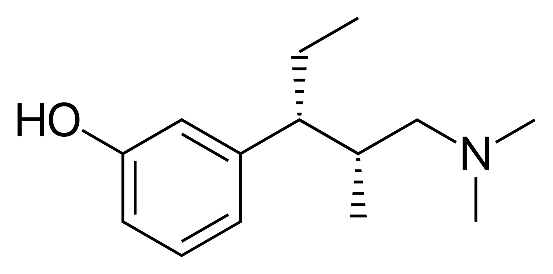
This case is focused on polymorphs, namely what are the standards for novelty and inventiveness when it comes to new crystal forms of a known drug? A recent Supreme People’s Court decision in China is illustrative of the way Chinese courts are thinking about polymorph patents in China. Grünenthal is a German pharmaceutical company and inventor of the opioid painkiller Pentadol[1]. Grünenthal has a Chinese patent (ZL 200580021661.1) with claims directed towards a new crystalline form of particular phenol hydrochloride (“tapentastat hydrochloride” and “Form A” in claim 1), processes for preparing tapentastat hydrochloride (claim 3), and processes for preparing Form A (claims 16 and 23). Form A is made by…